
The key to solving leaf yellowing isn’t just identifying the problem, but accurately reading the plant’s specific ‘symptom signature’ to avoid costly misdiagnosis.
- Nutrient mobility determines whether chlorosis appears on older leaves (mobile nutrients like magnesium) or new growth (immobile nutrients like iron).
- Field-level patterns—random patches versus uniform yellowing—are critical clues to differentiate between viral diseases and widespread nutrient deficiencies.
Recommendation: Adopt a systematic, ‘differential diagnosis’ approach. Observe the precise location and pattern of yellowing across the plant and field before committing to a treatment plan.
The sight of yellowing leaves spreading across a promising winter wheat crop is a universal concern for arable farmers. The immediate assumption is often a nitrogen shortfall, leading to a quick, and sometimes incorrect, application of fertiliser. While nitrogen is a frequent culprit, this knee-jerk reaction overlooks a crucial fact: chlorosis, the technical term for this yellowing, is not a single problem but a symptom with many potential causes. It’s the plant’s way of signalling distress, but the message is often more nuanced than it first appears.
The true key to an effective and cost-efficient solution lies not in guessing, but in a methodical process of differential diagnosis. What if the real issue is not a lack of nutrients in the soil, but the plant’s inability to access them due to incorrect pH? Or what if the yellowing is a sign of a viral infection, where fertiliser would be completely ineffective? This guide is built to transform you from a concerned observer into a crop detective. We will move beyond the generic advice and equip you with the knowledge to read the specific patterns and signatures of chlorosis.
By understanding the language of the leaves—distinguishing between symptoms on old versus new growth, recognising field-level distribution patterns, and appreciating the hidden role of soil chemistry—you can pinpoint the root cause. This diagnostic approach allows for targeted interventions that save money, protect the environment, and ultimately secure your yield potential. It’s about working smarter, not just applying more.
Table of Contents: A Diagnostic Guide to Wheat Chlorosis
- Why Magnesium Deficiency Hits Old Leaves While Iron Hits New Growth?
- How to Use Leaf Tissue Analysis to Confirm a Chlorosis Diagnosis?
- Virus vs Deficiency: Which Causes Your Barley Yellows?
- The pH Mistake That Mimics Nitrogen Deficiency in Oilseed Rape
- Foliar vs Granular: When to Intervene for Rapid Greening?
- The Micronutrient Interaction That Blocks Iron Uptake in Spring
- Why Rhizobia Bacteria Need Molybdenum to Fix Nitrogen Efficiently?
- How to Access Bio-Available Nutrients Locked in UK Chalk Soils Without Adding Lime?
Why Magnesium Deficiency Hits Old Leaves While Iron Hits New Growth?
The first and most powerful clue in diagnosing chlorosis is its location on the plant. This is dictated by a principle called nutrient mobility. Nutrients are transported through the plant’s vascular system, the phloem. Some are ‘mobile,’ while others are ‘immobile.’ Magnesium (Mg) is a classic example of a phloem-mobile nutrient. When the plant senses a shortage, it can scavenge Mg from older, less productive leaves and redirect it to new, high-priority growth at the top. As a result, deficiency symptoms—typically a striped or mottled interveinal yellowing—appear first and most severely on the older, lower leaves.
This mobility is a highly efficient survival mechanism for the plant. In fact, research demonstrates that 7-11% of absorbed Mg can be exported from treated leaves within just 24 hours to support other parts of the plant. This is why the Plant and Soil Journal notes, “Magnesium deficiency symptoms are typically associated with interveinal chlorosis particularly on older leaves as Mg is readily phloem-mobile and, therefore, retranslocated.”
In stark contrast, Iron (Fe) is a phloem-immobile nutrient. Once it’s incorporated into leaf tissue, it’s locked in place. The plant cannot move it from old leaves to new ones. Consequently, when iron is scarce, the new leaves emerging at the top of the plant are starved from the start. They will show a uniform pale yellow or even white chlorosis, while the older leaves remain green. Simply observing whether the yellowing starts from the top or the bottom of your wheat plant provides a critical first step in your differential diagnosis, immediately narrowing the list of potential culprits.
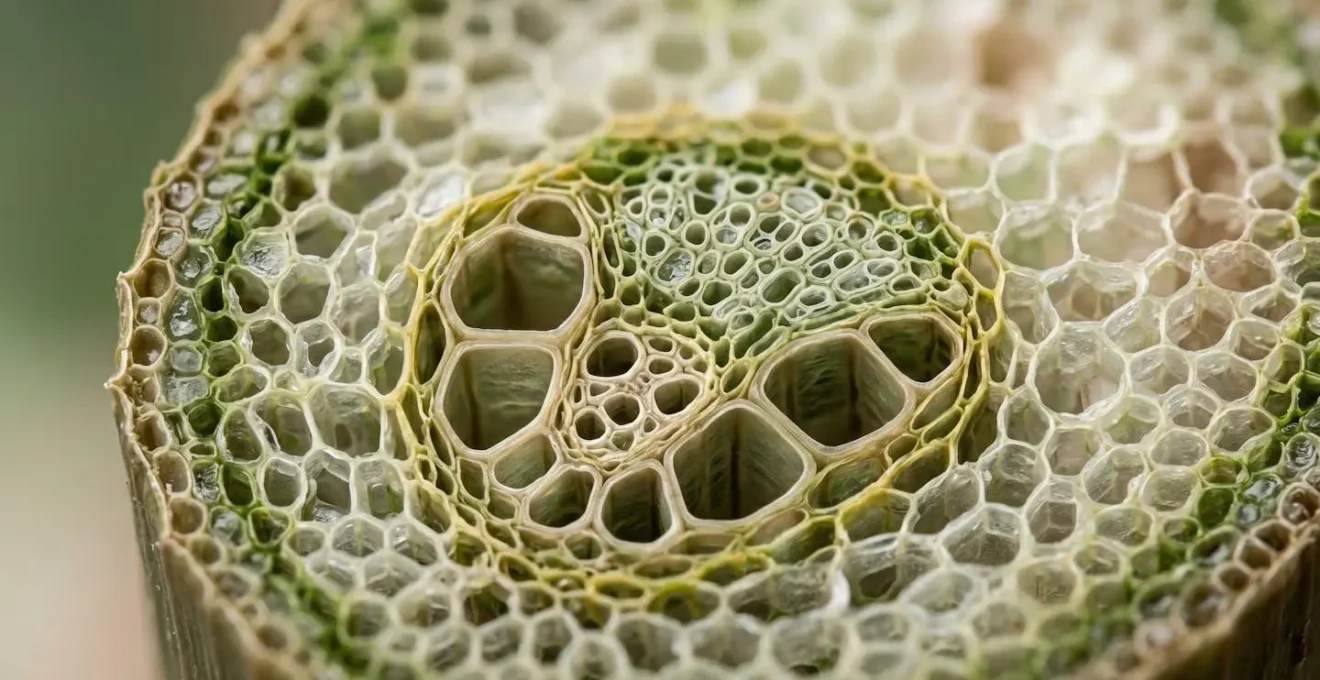
The intricate structure of the plant’s vascular tissue, shown above, is the highway for this nutrient transport. Understanding the traffic rules—which nutrients can move freely and which are on a one-way trip—is fundamental to interpreting the plant’s distress signals correctly. Seeing yellow on old leaves points you toward mobile nutrients like magnesium, nitrogen, or phosphorus, while yellow on new leaves points squarely at immobile ones like iron, manganese, or zinc.
How to Use Leaf Tissue Analysis to Confirm a Chlorosis Diagnosis?
While visual symptoms provide powerful directional clues, a leaf tissue analysis is the definitive step to confirm a nutrient deficiency and quantify its severity. It acts as the ‘blood test’ for your crop, moving beyond what’s in the soil to reveal what the plant has actually managed to absorb. Simply testing the soil can be misleading; a nutrient might be present but locked up and unavailable to the plant. Tissue testing bypasses this ambiguity, giving you a precise snapshot of the plant’s nutritional status at a critical growth stage.
A modern lab analysis does more than just report individual nutrient levels. Advanced methods like the Diagnosis and Recommendation Integrated System (DRIS) focus on the ratios between nutrients. As research on the system explains, “DRIS compares elemental ratio indices of elements with the established norms from an optimum high-yielding population.” This is crucial because nutrient balance is as important as absolute levels. For example, a high level of potassium can inhibit the uptake of magnesium, and the tissue test will reveal this antagonistic relationship.
One of the most practical applications of this is diagnosing sulfur deficiency, which often mimics nitrogen yellowing. A key metric that labs use is the nitrogen-to-sulfur (N:S) ratio. According to diagnostic standards, N:S ratios of 18:1 or greater are a strong indicator of sulfur limitation, even if nitrogen levels appear adequate. This simple ratio provides an actionable insight that visual inspection alone might miss. To get reliable results, however, correct sampling is non-negotiable.
Your Action Plan for Accurate Leaf Tissue Sampling
- Identify the Growth Stage: Take samples at the correct, recommended growth stage for winter wheat (e.g., tillering or stem extension). Nutrient norms are calibrated for specific stages.
- Select the Correct Leaf: Sample the most recently matured, fully expanded leaf (often the ‘Y-leaf’ or flag leaf later in the season). This is the plant’s active metabolic hub.
- Gather a Representative Sample: Collect at least 30-50 individual leaves from a wide, random zigzag pattern across the affected area. Avoid field edges, headlands, and dusty plants.
- Separate Good vs. Bad: If you have a clear ‘good’ area and ‘bad’ area in the field, take two separate samples. Submitting both for comparison provides the lab with an invaluable diagnostic baseline.
- Handle and Ship Correctly: Place leaves in a paper bag (not plastic, which causes sweating and degradation) and send them to the lab immediately. If there’s a delay, refrigerate the sample.
Virus vs Deficiency: Which Causes Your Barley Yellows?
Not all yellowing is a nutritional problem. Viral infections, particularly Barley Yellow Dwarf Virus (BYDV), are a significant cause of chlorosis in cereals and can be easily mistaken for a nutrient deficiency. As highlighted in *Frontiers in Plant Science*, the symptoms of viral disease can be confused with a wide range of other issues, including “nutrient deficiency or any other environmental stress.” Wasting expensive fertiliser on a viral infection is a common and costly mistake. The key to differentiating between the two lies in observing the symptom signature at the field level.
Nutrient deficiencies, especially those related to soil properties like pH or texture, tend to appear in more uniform, widespread patterns. You might see yellowing across an entire field, on a specific soil type, or along slopes and knolls where soil is thinner. The transitions are often gradual. Viral infections, on the other hand, are typically spread by insect vectors like aphids. Aphid movement is random, leading to a distinctively patchy distribution of the disease. You will often see scattered ‘hotspots’ of stunted, yellowed plants among an otherwise healthy green crop. These patches can appear circular or irregular and are sharply defined.

The visual evidence in the field, as depicted above, is your strongest diagnostic tool. The random scattering of affected plants is the classic calling card of a vector-borne disease and is fundamentally different from the more structured patterns of a nutrient issue. In addition to the field pattern, individual plants infected with BYDV often show a brighter, almost canary-yellow or reddish-purple discolouration, and growth is typically severely stunted compared to neighbouring healthy plants. If you see this combination of random patchiness and severe stunting, a viral cause should be your primary suspect.
The pH Mistake That Mimics Nitrogen Deficiency in Oilseed Rape
Often, the problem isn’t a lack of nutrients in the soil, but a chemical barrier preventing the plant from absorbing them. Soil pH is the master variable controlling nutrient availability. A common diagnostic error, particularly in oilseed rape and other cereals, is mistaking the effects of low soil pH (acidity) for a simple nitrogen deficiency. While the plants appear pale and stunted, the root cause is a toxic soil environment, not a lack of nitrogen.
As soil pH drops, the chemistry of the soil solution changes dramatically. In highly acidic conditions, naturally occurring aluminium, which is harmless in neutral soils, becomes soluble and toxic to plant roots. This is a widespread issue, as research indicates that soils with a pH of 5.5 or lower can trigger Al³⁺ toxicity, a problem affecting around 30% of the world’s land area. This toxic aluminium prunes the plant’s root system, severely restricting its ability to explore the soil for water and all nutrients, including nitrogen.
The above-ground symptoms—pale, weak growth—look exactly like nitrogen deficiency. A farmer might apply more N fertiliser, but since the damaged root system cannot absorb it effectively, the application has little to no effect. It’s a classic case of treating the symptom, not the disease. The only true solution is to correct the underlying soil acidity by applying lime to raise the pH. This ‘locks up’ the aluminium, making it non-toxic and allowing the plant’s roots to recover and function properly. A simple soil pH test is therefore one of the most cost-effective diagnostic tools available, preventing the misapplication of fertiliser and addressing the true limiting factor.
This principle also applies at the other end of the spectrum in high-pH, alkaline soils, where micronutrients like iron and manganese become unavailable. In both cases, the pH is creating a deficiency that cannot be solved by simply adding more of the nutrient that appears to be lacking. Understanding your soil’s pH is foundational to any nutrient management plan.
Foliar vs Granular: When to Intervene for Rapid Greening?
Once you’ve diagnosed a nutrient deficiency, the next decision is how to intervene. The choice between a granular, soil-applied fertiliser and a liquid, foliar-applied spray depends entirely on your objective: are you fixing a long-term soil issue or providing immediate ‘first aid’ to a stressed crop? Granular fertilisers are the foundation of soil fertility. They are designed to build and maintain the nutrient bank in the soil over the long term. They are best applied pre-drilling or in early spring to ensure a season-long supply. However, they are slow-acting and depend on soil moisture and microbial activity to become available to the plant.
Foliar fertilisers, by contrast, are a rapid-response tool. When a tissue test confirms a deficiency mid-season, a foliar spray delivers a small, targeted dose of nutrients directly to the leaves, where they are absorbed quickly. This bypasses any potential soil issues like pH lock-up or dry conditions that might be hindering root uptake. It’s the perfect tool for a quick green-up and to correct a deficiency that is actively limiting yield potential. It is not, however, a substitute for good soil fertility; it’s a tactical intervention, not a strategic overhaul.
The ability to distinguish between deficiencies is critical for timing. For instance, as noted by Bayer Crop Science, “Sulfur deficiency mimics N deficiency with light green to yellowish leaves. However, the whole plant is pale with S deficiency and the youngest leaves have more chlorosis than with N deficiency.” Recognising this subtle difference allows a grower to apply a fast-acting foliar sulfur product to immediately address the problem, rather than incorrectly applying more soil nitrogen. Foliar application is the intervention of choice when speed is critical or when you know soil conditions are the limiting factor.
The decision framework is simple: use granular products to feed the soil for the long term, and use foliar sprays to feed the plant for the short term. An integrated nutrient management plan uses both tools strategically to ensure the crop has what it needs at every stage of its growth cycle.
The Micronutrient Interaction That Blocks Iron Uptake in Spring
Plant nutrition is a delicate balancing act. Sometimes, a deficiency of one nutrient is not caused by a lack of it in the soil, but by an excess of another. This phenomenon, known as antagonistic interaction, is a common source of diagnostic confusion. An excess of one nutrient can compete with another for uptake by the plant’s roots, effectively blocking it out. One of the most classic antagonistic relationships in cereal crops is between phosphorus and zinc/iron. Applying high rates of phosphorus fertiliser, especially in cool, wet spring soils, can induce a zinc or iron deficiency, even if levels of those micronutrients in the soil are technically adequate.
This again ties back to the principle of nutrient mobility. As UF/IFAS Extension points out, immobile nutrients include “Iron (Fe), Zinc (Zn), Manganese (Mn), [and] Boron (B).” Because these nutrients are immobile, deficiency symptoms show up in the new growth first. If you see yellowing on the new leaves of your wheat after a heavy phosphate application, antagonism should be a primary suspect.
Environmental conditions can also create temporary antagonistic effects. Cool, wet soils in the spring are notorious for limiting nutrient uptake, particularly for magnesium and phosphorus. The plant’s metabolic activity slows, and roots struggle to function efficiently. Field observations often reveal that wheat is unable to absorb adequate Mg in cool, wet springs, especially in soils with a lower pH. This isn’t a true soil deficiency, but a temporary, weather-induced problem. In this scenario, a foliar application of the deficient nutrient is the perfect remedy, bypassing the cold, sluggish soil and delivering nutrition directly to the plant when it needs it most.
Understanding these interactions is vital for a correct diagnosis. Before concluding that a nutrient is missing from your soil, consider whether an excess of another nutrient or adverse environmental conditions could be the real culprit. This mindset prevents unnecessary and ineffective fertiliser applications, targeting the true source of the problem.
Why Rhizobia Bacteria Need Molybdenum to Fix Nitrogen Efficiently?
While often overlooked, the micronutrient molybdenum (Mo) plays a disproportionately large role in the nitrogen cycle, the most critical process for plant growth. Its importance is not in the plant itself, but in the symbiotic bacteria that provide the plant with nitrogen. Legumes, such as peas, beans, and clover, which are often used in rotation with wheat, form a partnership with Rhizobia bacteria. These bacteria live in nodules on the plant’s roots and perform the seemingly magical task of converting atmospheric nitrogen gas (N₂) into a form the plant can use—a process called nitrogen fixation.
This entire process hinges on a single, highly specialised enzyme called nitrogenase. Molybdenum is the essential metallic cofactor at the heart of this enzyme. Without sufficient molybdenum, the Rhizobia bacteria cannot produce functional nitrogenase. As a result, the nitrogen-fixing process grinds to a halt. The legume crop is starved of nitrogen, showing classic symptoms of N deficiency (pale, yellowing leaves), and fails to enrich the soil with nitrogen for the following wheat crop.
A molybdenum deficiency, therefore, creates a nitrogen deficiency by proxy. This is especially relevant in acidic soils, where molybdenum availability is naturally low. A farmer might see a poorly performing clover or bean crop, diagnose it as an N deficiency, and apply nitrogen fertiliser. This is not only inefficient but misses the root cause. A very small, inexpensive application of molybdenum could have restored the bacteria’s function, allowing the system to produce its own nitrogen for free.
Because of its critical role in certain crops, agronomists pay close attention to it. As North Carolina’s Agronomic Services state, “Molybdenum is a micronutrient that is important in certain crops. It is automatically included in the standard analysis for Brassicas, alfalfa, and spinach.” This highlights its importance in rotational planning, ensuring that a lack of this tiny micronutrient doesn’t cripple the engine of your farm’s nitrogen cycle.
Key Takeaways
- Symptom location is key: Yellowing on old leaves indicates a mobile nutrient issue (like Mg), while yellowing on new growth points to an immobile nutrient (like Fe).
- Field patterns tell a story: Uniform yellowing often signals a soil-based nutrient problem, whereas random, scattered patches are a classic sign of a vector-borne viral disease.
- Soil pH is the master controller: Both low (acidic) and high (alkaline) pH can lock up essential nutrients in the soil, causing deficiency symptoms even when the nutrient is physically present.
How to Access Bio-Available Nutrients Locked in UK Chalk Soils Without Adding Lime?
The diagnostic framework we’ve discussed is universal, but its application must be tailored to specific soil types. For farmers on the chalk and limestone soils common in parts of the UK, the primary challenge is not acidity, but high pH (alkalinity). These soils, rich in calcium carbonate, can have a pH of 7.5 or higher. While this provides excellent soil structure, it creates a major chemical headache: the lock-up of essential micronutrients.
In high-pH environments, micronutrients like iron (Fe), manganese (Mn), and zinc (Zn) become insoluble and unavailable for plant uptake. The soil may contain vast reserves of these elements, but they are not in a bio-available form. The result is induced chlorosis, most commonly seen as interveinal yellowing on the new leaves of the crop, as these are all immobile nutrients. Adding more standard fertiliser is futile, and applying lime—the solution for acidic soils—would be disastrous, pushing the pH even higher and worsening the lock-up. Here, the diagnosis is clear: the problem is not a lack of nutrients, but a lack of access.
The solution requires bypassing the soil chemistry. The most effective intervention is targeted foliar application. Spraying liquid formulations of the deficient micronutrients directly onto the crop’s leaves allows for rapid absorption, providing immediate relief and protecting yield potential. For manganese deficiency, a common issue on these soils, regular foliar Mn sprays throughout the spring are a standard part of a successful management program. Another strategy is the use of chelated micronutrients. Chelates are compounds that protect the nutrient from being locked up by the soil, keeping it available for root uptake for longer. While more expensive, they can be a valuable tool when applied with seed or in early granular applications.
Managing high-pH soils is a perfect example of the diagnostic mindset in action. By understanding the underlying chemical barrier, growers can avoid incorrect inputs and choose targeted, effective solutions that work with their specific environment, not against it.
To effectively manage crop health and boost profitability, the next logical step is to apply this diagnostic framework to your own fields. Start by systematically observing symptom patterns and soil characteristics before reaching for the sprayer, turning observation into your most powerful management tool.